SharePoint Quality Management Solutions
Ready-to-use Modules & Customizations For Your QMS
Transform your Quality Management System with a custom SharePoint-based solutions that mirrors your company's existing processes; there is no need to adapt to a rigid tools. We digitize and optimize what already works, ensuring your QMS becomes smarter, faster, and fully compliant.
Own Your System.Control Your Data.Scale on Your Terms.
1-to-1 Process Digitalization
Ready-made Modules
We offer ready-made QMS modules that can be implemented into QMS right away.
Enterprise-Grade Security
The entire system is deployed to your company SharePoint environment, secure Microsoft infrastructure.
1-to-1 Process Alignment
We also replicate your current workflows exactly where needed, adapting the system to you, not the other way around.
Full Ownership
You control the data and the environment with no vendor lock. Expand your system with us or other partners.
Fully Compliant
All components are designed in line with ISO 13485, FDA 21 CFR Part 820 and Part 11.
Validated Solution
We provide validation reports and detailed technical documentation for each QMS component.
User Friendly and Compliant eQMS
Digitized Working Environment
Your digital QMS should do more than store documents, it should actively drive compliance, efficiency, and visibility across all quality processes.
Fully Automated QMS
Built on Microsoft SharePoint and Power Platform, our solution unites automation, traceability, and security to simplify every aspect of quality management, from document control and workflows to real-time insights and audit readiness.
Customized Modules for Your Specific Needs
We customize our modules to mirror your existing processes where needed. The system strikes a balance between easy deployment and the customization you need to keep your internal processes unchanged.

From document creation to real-time insight. Your QMS, streamlined, automated, comfortable.
Customizable eQMS Modules and Solutions
We provide ready-made eQMS modules that cover the most common quality management needs in the healthcare industry. We can also create customized modules from our proven building blocks, enabling fast development and leveraging the infrastructure and experience we have already built.
Automating & Digitizing Your Processess and Workflows
We also offer building components from which we create any application exactly according to your requirements.

Your QMS & Your Processes, streamlined and automated.
Documentation Automation and Management
Every process starts with structured templates aligned with ISO 13485, ISO 9001, or FDA 21 CFR 820.30.
Documents are created and linked directly in SharePoint, ensuring consistency, integrity, and full traceability.
- 1 to 1 Document Templates
- Automated PDF Conversion
- Secure SharePoint Storage
- Version Control & Audit Trails

Compliance & Security
Our QMS solution is developed following ISO 80002-2 principles for computer system validation and aligned with major international standards.
Standards Compliance
- ISO 13485
- ISO 9001
- FDA 21 CFR Part 820 and Part 11
- ISO 27001
Electronic Signatures & Audit Trails
Built-in accountability and compliance readiness.
Secure & Controlled Data
No vendor lock-in, no third-party storage; everything stays within your Microsoft environment.
Validated & Audit-Ready
Every function is tested and documented for regulatory assurance.

Quality Management System for Medical Devices

Electronic Signatures & Audit Trails

In Vitro Diagnostic Regulation

Medical Device Regulation

Health Insurance Portability and Accountability Act

General Data Protection Regulation

Good Practices
Flexible Pricing for Custom Module Development
Choose the solution that best fits your organization’s process. Whether you need a simple, structured solution or a fully customized, advanced system, our flexible pricing ensures you only pay for what you need.

Essential Plan
"Get Started with a Simple, Structured Solution"
- Streamline approvals with a lightweight, structured approach
- Perfect for teams with basic validation needs
- Simple workflow, essential compliance
4 000 - 6 000 EUR
Advanced Plan
"Take Your Validation Process to the Next Level"
- More templates, more control, more efficiency
- Improve collaboration and process transparency
- For organizations with mid-level process complexity
7 000 - 9 000 EUR
Enterprise Plan
"Fully Customized for Ultimate Control"
- A fully tailored validation solution built for your organization
- Custom roles, unlimited workflows, and deep process automation
- Full compliance, full scalability, full control
10 000+ EUR
Custom pricing available
Every organization and every QMS journey is different. If your goals, timelines, or technical complexity don’t fit a standard package, we offer custom pricing aligned with your specific needs, scope, and regulatory context.


How We Build SharePoint eQMS Solution
Structured and scalable approach ensuring compliance, and adapting to your business needs
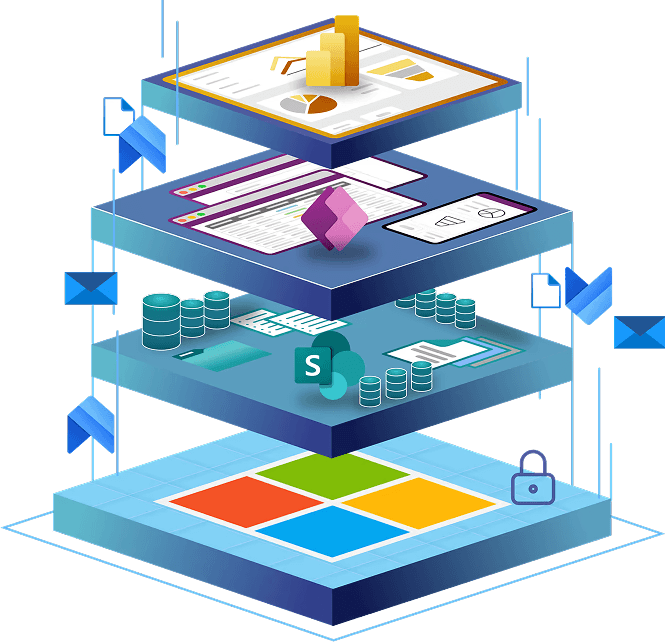
SharePoint Foundation and Platform Expansion
Every solution begins with a robust Microsoft foundation, combining SharePoint and the Power Platform to deliver a secure, compliant, and automated quality environment.

SharePoint
Establishes centralized, version-controlled document storage with retention policies, access management, and full traceability from day one.
We extend your SharePoint foundation with the Microsoft Power Platform to transform manual processes into automated, data-driven workflows.

Power Apps
Build intuitive, role-based applications that digitize quality processes like CAPA, training, or audits.

Power Automate
Automate approvals, notifications, and routine tasks while maintaining full audit trails.

Power BI
Deliver real-time insights, compliance dashboards, and management reports for smarter decision-making.

Microsoft Teams
Enable instant collaboration, task updates, and communication without leaving your workflow environment.

Microsoft Outlook
Integrate approvals, alerts, and task assignments directly into your inbox.
Scalable Customization, Integration and Extensions
Once the core system is live, we enhance and extend it further, tailoring dashboards, workflows, and reports to your exact needs.
We can integrate existing systems, platforms and tools you use (like ERP, CRM, PLM) and connect them with your QMS to create a unified compliance ecosystem without disrupting ongoing operations.
Beyond Power Platform – Advanced Custom Development
When your processes require functionality beyond standard tools, we use our software engineering expertise to deliver custom features and integrations.
Using SharePoint Framework (SPFx), Graph API, Azure Functions, React, C# and custom APIs, we extend your QMS with precision, enabling deep customization, seamless integrations, and future scalability.
AI-Enabled Quality Management Processes
We embed AI directly into SharePoint-based eQMS workflows to support automated checks for completeness, consistency, and logical alignment across QMS documentation. This helps identify gaps, inconsistencies, and potential compliance risks early, enabling faster reviews, more reliable change management, and improved traceability—while keeping human oversight firmly in control.
Secure, Enterprise AI Architecture
Our solutions use Microsoft Copilot, Azure AI, and Retrieval-Augmented Generation (RAG) to build company-specific knowledge bases grounded in your own data. By integrating SharePoint content and validated quality records, we deliver contextual, role-based AI assistance. We can extend functionality with external agents such as n8n or custom components, all operating within your environment—fully owned, validated, monitored, and governed by your organization.
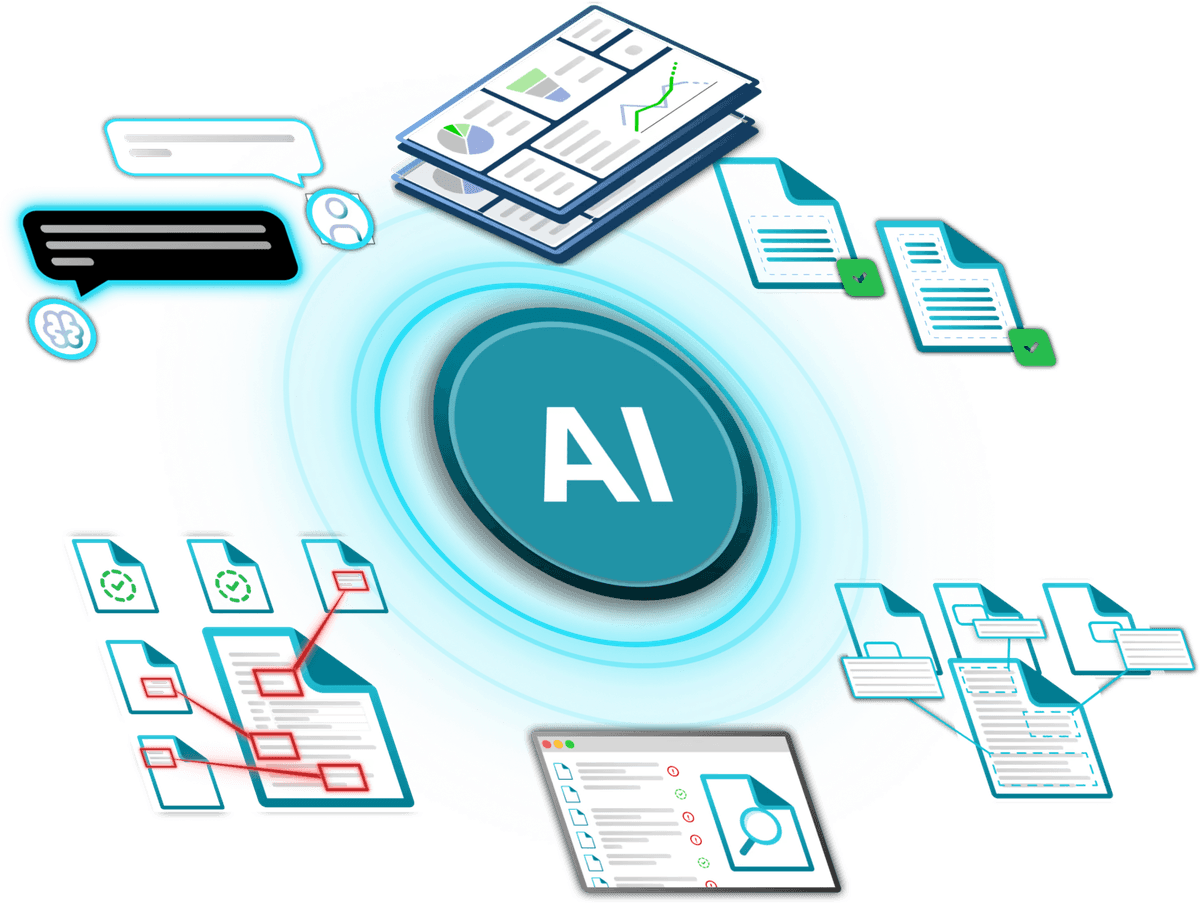
Validated & Future-Ready Solution
Our eQMS implementations are designed and documented following ISO 80002-2 guidelines for computerized system validation, ensuring full confidence in regulatory compliance and system reliability.
Continuous Compliance & Adaptability
Compliance isn’t a one-time milestone, it’s a continuous commitment.
Our systems are equipped with electronic signatures, audit trails, and documentation control, ensuring ongoing regulatory alignment with ISO 13485, 21 CFR Part 11, and other standards.
We design every solution to evolve with your organization, ready for future regulatory changes, process updates, or business growth.
The Result
A fully owned, compliant, and future-ready QMS that delivers:
- Consistency across all documents and processes
- Digitized workflows that eliminate manual inefficiencies
- Automated routine tasks with reduced risk of error
- Enhanced compliance & security aligned with ISO and FDA standards
- Scalability to grow, extend, and integrate with new tools and systems
At QMLogic, we build QMS solutions that grow with you; combining regulatory rigor with the flexibility of SharePoint and Power Platform to ensure long-term value and adaptability.

Enhancing your QMS; not replacing it.
Key Benefits of SharePoint and Microsoft 365 QMS Solutions
Full Ownership & Control
No vendor lock-in or external hosting.
Your data stays entirely within your Microsoft 365 environment. You retain full control, access, and ownership, ensuring long-term independence and data sovereignty.
Secure & Enterprise-Grade
Built within Microsoft’s infrastructure, your QMS benefits from enterprise-level authentication, data protection, and scalability. Security and compliance features are embedded by default.
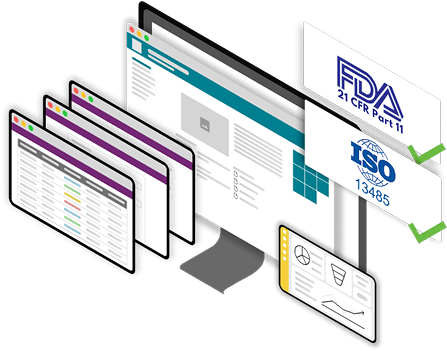
Compliant by Design
Our solutions are built in alignment with ISO 13485, FDA 21 CFR Part 11, and related quality and data integrity standards. Every workflow, form, and document is designed to support audit readiness and regulatory validation from day one.
Fully Adaptable
Mirrors your internal processes, approval flows, and documentation structure. We design Power Apps and Automate workflows that evolve with your organization — not against it.
Seamlessly Integrated
Works natively with Microsoft 365 Tools like Teams, Outlook, or OneDrive, connecting communication, data, and tasks in one ecosystem. No more disconnected tools; just one connected environment that keeps quality moving.
Cost-effective & Scalable
Built on the tools you already own, without recurring third-party subscription fees. You can expand, automate, and scale as your organization grows, efficiently and sustainably.
Automated & Efficient
Reduce manual work and repetitive steps with automated approvals, notifications, and task tracking. Real-time visibility ensures faster decisions, consistent quality, and fewer compliance bottlenecks.
Built on Microsoft. Designed for Compliance. Tailored to You.
Testimonials | Expertise in Process Digitalization
Vaclav did support our journey to become ISO 13485 certified with relentless work put into great processes, great communication and detailed and to the point conversations with the teams, individual members - but also our external auditors. He was of great support to move our QMS to the next level. He was the key person behind the move away from paper / scan based documents towards a digital setup.If you are in need for a structured person with great communication skills, a good understanding of the regulatory environment, I can only recommend to reach out to Vaclav. Lucky you if he's not booked ;-) Besides all the success he's an enjoyable and humble character. Great experience to having worked with him.
Vaclav and I worked together on risk management activities of medical device development projects. His ability to quickly grasp the challenges and to accomplish a task/project is remarkable. Additionally, his ability to propose and develop software solutions to improve projects’ efficiency is commendable. I find Vaclav to be flexible, dependable, and quality centric. Hence, I really enjoyed working with Vaclav and looking forward to associate with him again.
Questions & Answers
Yes, we can help you choose the right solution for you, either because you just decided to leave your current vendor, or you are first establishing a quality management system.
Together, we will analyze your current situation and define the requirements that are most important to you, and we can advise you which quality management system solution to choose.
We have the most experience in building quality management systems around the Atlassian or Microsoft suite, but we can evaluate any other software solution that would fit your needs.
For specific requirements and organizational setups, we also provide custom-made software solutions, fully developed by us.
Get consultancy for free
Ask anything you need to know about Medical Software, CE certification or MDR.
- Learn how we can streamline your processes.
- Schedule a demo to see the solution in action.
- Request customization for your QMS and SharePoint.

